Organisms that require oxygen to grow and survive are called aerobes, and the short answer is this: they are bacteria that require oxygen to grow are called they need oxygen because they use it as the final electron acceptor in aerobic respiration, the process that generates the energy they need to live. Without oxygen, that energy-producing pathway shuts down, and so does their ability to grow. But 'requiring oxygen' is not a single, binary thing. There is a whole spectrum of oxygen requirements among microorganisms, and understanding where a particular organism falls on that spectrum is one of the most practically useful things you can know, whether you are culturing bacteria in a lab, storing food safely, or studying for a microbiology exam. an organism that cannot grow without oxygen is a an. bacteria that grow in oxygenated environments are referred to as. a halophile would grow best in quizlet
Organisms That Require Oxygen to Grow and Survive

What 'Oxygen-Requiring' Actually Means
When we say an organism requires oxygen, we mean it depends on molecular oxygen (O2) to carry out its metabolic processes. More specifically, these organisms use oxygen as the terminal electron acceptor at the end of the electron transport chain during aerobic respiration. Think of the electron transport chain as a series of falling dominoes that release energy with each step. Oxygen is the last domino, the final acceptor that catches all those electrons and combines with hydrogen to form water. If oxygen is not there, the chain cannot complete, and the cell cannot generate the ATP (energy currency) it needs at the rate required for growth and reproduction.
This is fundamentally different from organisms that simply tolerate oxygen or that can switch to other strategies when oxygen disappears. A true oxygen-requiring microbe is locked into aerobic respiration as its primary energy strategy. Remove the oxygen, and you remove the organism's ability to thrive. That biological dependency is what makes oxygen level such a powerful and reliable tool for classifying and identifying microorganisms in the lab.
The Oxygen Requirement Spectrum: Four Categories You Need to Know

Microbiologists do not just sort organisms into 'needs oxygen' and 'does not need oxygen.' There are at least four meaningful categories, and knowing the difference matters a lot in both lab settings and real-world applications.
Obligate Aerobes
These are the strict oxygen-requirers. An obligate aerobe cannot grow without molecular oxygen. It has no fallback energy pathway and will die or become dormant in anaerobic (oxygen-free) environments. Mycobacterium tuberculosis, the bacterium responsible for tuberculosis, is a classic example. It preferentially colonizes the upper lobes of the lungs, which are the most oxygen-rich regions of the respiratory system. That is not a coincidence; it is biology playing out in real time.
Microaerophiles

Microaerophiles require oxygen but at concentrations lower than the roughly 21% found in normal atmospheric air. High oxygen concentrations actually inhibit or damage them. Campylobacter jejuni, a leading cause of foodborne illness worldwide, is a microaerophile that thrives at around 5% oxygen. This matters practically: if you are culturing Campylobacter in a standard aerobic incubator, you will get poor or no growth, and you might incorrectly conclude the sample is negative.
Facultative Anaerobes
Facultative anaerobes are the metabolic multitaskers. They prefer oxygen and grow best when it is available (because aerobic respiration is more efficient), but they can switch to fermentation or anaerobic respiration when oxygen disappears. Escherichia coli is the textbook example. Facultative organisms are not strictly oxygen-requiring, but they definitely use it opportunistically. Many common pathogens, including Staphylococcus aureus and most Enterobacteriaceae, fall into this group.
Obligate Anaerobes

These organisms not only do not need oxygen, they are actively harmed by it. Obligate anaerobes like Clostridium botulinum or Bacteroides fragilis have little to no superoxide dismutase or catalase activity. These are the enzymes that neutralize the toxic reactive oxygen species (like superoxide radicals) that form when oxygen is present. Without those enzymatic defenses, even brief oxygen exposure can be lethal. This is the polar opposite of an obligate aerobe, and it has major implications for sample handling in clinical microbiology. If you are collecting a wound sample suspected of anaerobic infection, oxygen exposure during transport can kill the very organisms you are trying to identify.
| Category | Oxygen Requirement | Growth Without O2 | Example Organisms |
|---|---|---|---|
| Obligate Aerobe | Requires O2 to grow | No | Mycobacterium tuberculosis, Pseudomonas aeruginosa |
| Microaerophile | Requires low O2 (2–10%) | No (or very poor) | Campylobacter jejuni, Helicobacter pylori |
| Facultative Anaerobe | Prefers O2, can grow without it | Yes | Escherichia coli, Staphylococcus aureus |
| Obligate Anaerobe | Cannot tolerate O2 | Yes (requires absence of O2) | Clostridium botulinum, Bacteroides fragilis |
Why Oxygen Powers Growth: The Aerobic Respiration Basics
To really understand why oxygen-requiring organisms depend so heavily on O2, you need a quick look at what aerobic respiration actually does. When an aerobe breaks down a glucose molecule through glycolysis and the citric acid cycle, it generates electron carriers (NADH and FADH2) that feed into the electron transport chain embedded in the cell membrane. As electrons move down the chain, protons are pumped across the membrane, building up a gradient. That gradient drives an enzyme called ATP synthase, which produces ATP, the cell's usable energy. At the very end of this chain, electrons are handed off to oxygen, which accepts them and bonds with hydrogen to form water.
The payoff is enormous: aerobic respiration yields approximately 36 to 38 ATP molecules per glucose, compared to just 2 ATP from fermentation. This is why organisms that use oxygen grow faster, reach higher cell densities, and outcompete non-aerobic organisms when oxygen is plentiful. Oxygen is not just useful to these organisms; it is the engine behind their competitive advantage. Strip it away, and an obligate aerobe has nothing left to run on.
How to Recognize Oxygen-Dependent Growth in the Lab

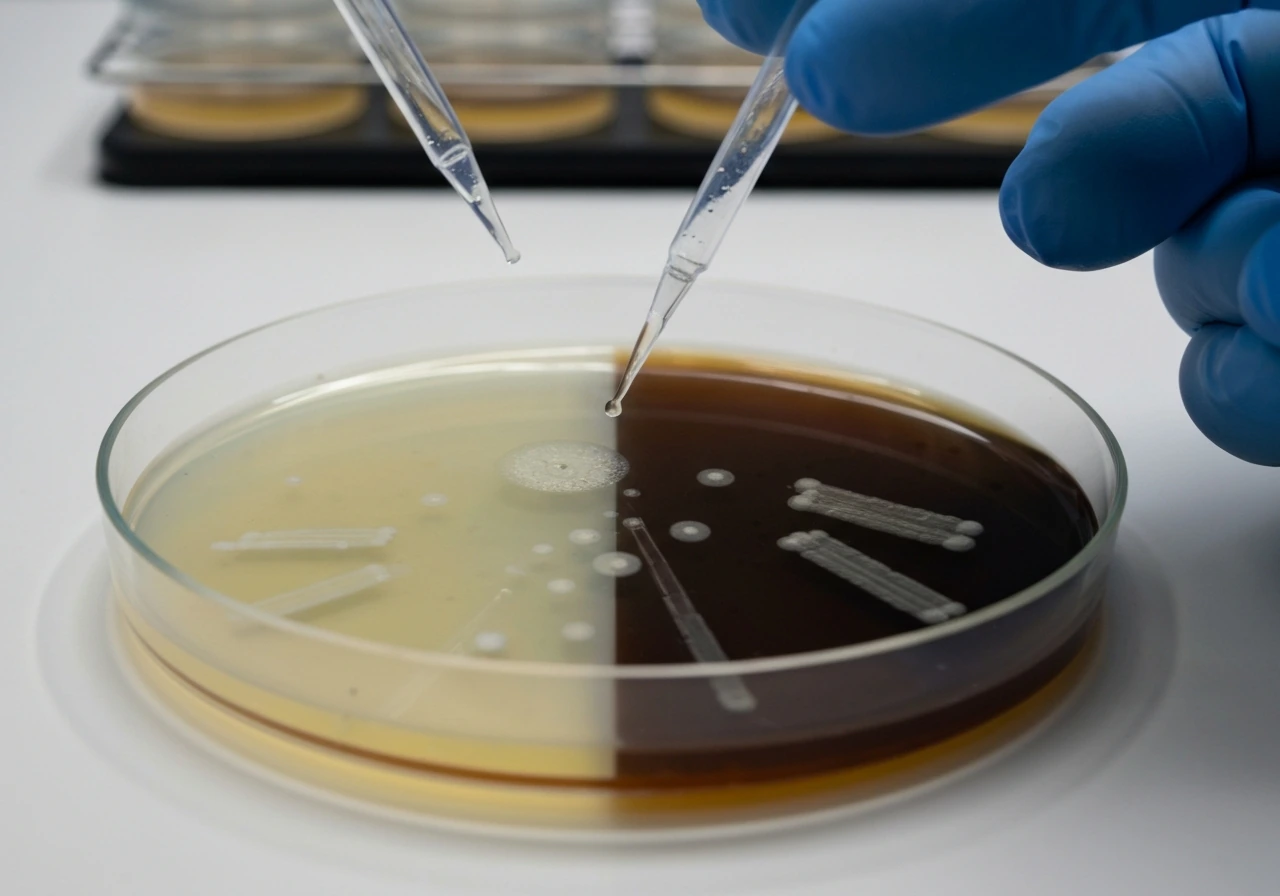
The most elegant and widely used tool for observing oxygen-dependent growth is the thioglycollate broth tube. Sodium thioglycollate reacts with dissolved oxygen, creating an oxygen gradient in the tube: high oxygen at the top near the surface, dropping to essentially zero at the bottom. When you inoculate a microorganism into this tube and incubate it, where the growth appears tells you exactly what the organism needs.
- Growth only at the top (aerobic zone): obligate aerobe
- Growth only at the bottom (anaerobic zone): obligate anaerobe
- Growth throughout the tube, but denser near the top: facultative anaerobe
- Growth in a band just below the surface (low-oxygen zone): microaerophile
This single tube result can tell you more about an unknown organism's fundamental biology than many other tests. It is one of the first steps in classifying an unfamiliar isolate. Beyond thioglycollate, you can also use catalase and superoxide dismutase (SOD) tests as supporting evidence. Obligate aerobes typically produce robust catalase activity (you will see vigorous bubbling when hydrogen peroxide is applied to a colony), because they need to neutralize the reactive oxygen species generated during normal aerobic metabolism. Obligate anaerobes, by contrast, will show little or no catalase activity, which is part of why oxygen is so toxic to them.
For organisms suspected to be microaerophiles, you will need a specialized incubation environment, typically a gas pack or controlled atmosphere incubator set to around 5% O2, 10% CO2, and 85% nitrogen. Normal incubators will not work. This is a common practical mistake that leads to missed or misidentified cultures.
Practical Steps for Growing or Avoiding Oxygen-Dependent Microbes
Whether you are trying to successfully culture an oxygen-requiring organism or trying to prevent one from growing in a food product, the principle is the same: control the oxygen environment deliberately. Here is how to approach it practically.
If You Are Culturing Oxygen-Requiring Organisms
- Identify the category first. Before you set up your culture, determine whether you are dealing with an obligate aerobe, microaerophile, or facultative organism. The thioglycollate tube result from a previous isolate, or prior knowledge of the organism, tells you what incubation setup you need.
- Use the right atmosphere. Obligate aerobes grow fine in a standard incubator open to room air (approximately 21% O2). Microaerophiles require a gas pack or CampyPak system to reduce O2 to 5–10%. Facultative organisms are flexible and will grow in either setup.
- Select appropriate media. Some oxygen-requiring organisms need specific nutrients alongside the right atmosphere. Blood agar supports many fastidious aerobes. Selective media like BCYE agar is used for Legionella, another aerobe, because it provides cysteine and iron that this organism cannot synthesize.
- Incubate at the correct temperature and duration. Oxygen requirement does not operate in isolation. Temperature, pH, and nutrients all interact. Most human pathogens are obligate aerobes or microaerophiles growing best at 35–37°C (body temperature), but some environmental aerobes prefer cooler conditions.
- Confirm results. After incubation, use catalase testing, Gram staining, and growth pattern observation to confirm your classification before drawing conclusions.
If You Are Handling Clinical or Environmental Samples
For clinical samples where you suspect an obligate aerobe, normal transport conditions (room air, relatively prompt delivery) are usually fine. The concern with oxygen actually runs the other way: if you suspect anaerobic organisms alongside aerobes in the same sample (which is common in polymicrobial infections), you need to use an anaerobic transport container for that portion of the specimen to avoid killing the anaerobes in transit. Failing to do this is one of the most common sources of false-negative anaerobic cultures in clinical labs.
In Food Safety Contexts

Controlling oxygen is one of the most powerful tools in food preservation. Vacuum sealing and modified atmosphere packaging (MAP) reduce oxygen, which selectively suppresses obligate aerobic spoilage organisms like Pseudomonas species (major causes of surface spoilage in meat and dairy). However, this same low-oxygen environment can potentially favor obligate anaerobes like Clostridium botulinum, which is why proper refrigeration and pH control remain essential even in vacuum-packed products. Reducing one risk can increase another if you do not account for the full picture.
Where Oxygen-Dependent Microbes Actually Show Up
Understanding where these organisms thrive in real-world contexts makes the biology feel much less abstract.
In the Human Body
The human body is a patchwork of oxygen environments. The lungs, skin surface, and upper respiratory tract are well-oxygenated, making them hospitable to obligate aerobes and microaerophiles. Mycobacterium tuberculosis targets the upper lung lobes specifically because oxygen tension is highest there. Helicobacter pylori, a microaerophile, colonizes the stomach lining, where the mucus layer creates the low-oxygen microenvironment it needs. Deep wound tissue, the colon, and the gingival crevice between teeth and gums are comparatively anaerobic, which is why these sites tend to harbor obligate anaerobes. Oxygen gradient is, in a very real sense, a map of which microbes live where in your body.
In Food and Kitchen Environments
Aerobic spoilage is the most common kind of food spoilage you encounter in everyday life. That slimy film on leftover chicken or the fuzzy mold on bread? Both are caused by aerobic organisms (bacteria and fungi, respectively) that thrive in the oxygen-rich environment of your kitchen and fridge. Pseudomonas fluorescens, a common refrigerator spoiler, is an obligate aerobe. Molds like Aspergillus and Penicillium are also obligate aerobes, which is why sealing food in airtight containers dramatically slows their growth. This is the practical application of aerobe biology that most people experience daily without realizing it.
In Cleaning and Hygiene Contexts
Most surface biofilms that develop in bathrooms, on countertops, and in drains are dominated by aerobic organisms because these are oxygen-exposed environments. Pseudomonas aeruginosa, a tough obligate aerobe, is notorious for forming biofilms in moist, oxygen-rich environments like shower drains and hospital equipment. Standard disinfectants work partly by disrupting the cell membranes and metabolic processes of these aerobic organisms. Some disinfectants can still leave oxygen conditions that allow certain organisms with oxygen dependence to persist organism that may even grow in certain chemical disinfectants is. Understanding their oxygen dependence helps explain why dry environments tend to have far fewer viable microbial contaminants than wet, aerated ones.
Common Misconceptions Worth Clearing Up
One of the most persistent misconceptions is the idea that if something is growing, oxygen must not matter. This comes up in lab troubleshooting when a culture grows fine but you assumed the organism was a strict aerobe. The explanation is almost always that the organism is actually a facultative anaerobe, not an obligate aerobe. Facultative anaerobes grow with or without oxygen, so their growth in a sealed container does not mean oxygen is irrelevant. It means the organism has already switched to fermentation. The key distinction is not just whether growth happens, but where it is most robust.
Another common confusion is between 'needing oxygen' and 'using oxygen.' Some people assume that because aerobic respiration produces more energy, all organisms would evolve to use it if they could. But obligate anaerobes did not simply fail to evolve aerobic respiration. Many of them evolved in environments that were oxygen-free for billions of years, before Earth's atmosphere became oxygenated. For these organisms, oxygen is a genuinely toxic environmental condition, not just an unused resource. Their enzyme systems are not equipped to handle it, and that is a fundamental biological fact, not a deficiency.
A third misconception involves life stage and microenvironment. Some organisms that are classified as aerobes based on lab testing actually encounter localized low-oxygen zones in their natural habitat. Biofilms are a perfect example: the outer layers of a thick biofilm might be aerobic, but organisms buried deep in the biofilm interior may experience near-anaerobic conditions. The organism's classification describes its optimal or required condition, not the only condition it ever encounters. This is also why you will sometimes see related articles discussing organisms that grow with or without oxygen, which are distinct from what we are covering here.
Finally, students sometimes wonder why certain pathogens are oxygen-loving when the interior of the human body is relatively low in oxygen. The answer is microenvironment. Many aerobic pathogens colonize oxygen-exposed surfaces (skin, lungs, nasal passages, and wounds open to air) rather than deep tissue. When an obligate aerobe like Pseudomonas aeruginosa does invade deeper tissue, it often struggles to survive there without the oxygen supply it needs, which is part of why the immune system can sometimes contain it to localized areas.
Putting It All Together
Organisms that require oxygen to grow and survive, true aerobes and microaerophiles, are defined by their dependence on oxygen as a terminal electron acceptor in aerobic respiration., true aerobes and microaerophiles, are defined by their dependence on oxygen as a terminal electron acceptor in aerobic respiration. That single biological requirement explains their growth patterns, their distribution in the body, their behavior in food, and the conditions you need to replicate in a lab to culture them successfully. The thioglycollate broth tube remains the most direct and practical way to classify an unknown organism's oxygen requirement, and the result guides everything from media selection to sample handling protocols.
What makes this topic so foundational in microbiology is that oxygen requirement does not exist in isolation. It connects directly to temperature, pH, and nutrient availability as interacting conditions that together determine whether a microorganism thrives or fails in any given environment. Whether you are troubleshooting a failed culture, studying for an exam, or making decisions about food storage at home, understanding oxygen requirements gives you a genuinely powerful lens for predicting microbial behavior.
FAQ
How can I tell whether an organism is obligate aerobe or microaerophile if it grows in my incubation setup?
Growth in an “aerobic” incubator alone is not enough. If you suspect microaerophily, repeat the test with controlled oxygen (for example, around 5% O2 using a gas pack). Obligate aerobes typically tolerate higher oxygen, while microaerophiles are inhibited at atmospheric levels.
What does “failure to grow” in an oxygen test actually mean?
It can mean more than oxygen dependence. The organism may be harmed by improper temperature, excessive CO2, wrong pH, insufficient nutrients, or delayed inoculation. If all other conditions match your protocol and only oxygen differs, then oxygen requirement becomes the most likely driver.
If an obligate aerobe grows only near the top of a thioglycollate tube, is that always conclusive?
It strongly suggests an obligate aerobe, but not always. Some organisms form pigments or biofilms and may appear as patchy growth. To confirm, pair the thioglycollate result with a catalase test and, ideally, an incubation under defined oxygen levels.
Can I use a sealed container at home to culture oxygen-requiring bacteria safely?
Not reliably, and also not safely to attempt with unknown microbes. For oxygen-requiring organisms, oxygen availability depends on headspace size, gasket tightness, and how long the culture is left. Without calibrated oxygen control, you can get misleading results.
Why do facultative anaerobes sometimes look like they are “needing oxygen” in a tube test?
Because they grow faster where oxygen is available. Even though they can switch to fermentation without O2, their growth rate and biomass are often highest near the top of an oxygen gradient. The key is the relative density and extent across the gradient, not just presence of growth.
How should specimen transport change if I suspect both aerobes and anaerobes?
Split the specimen handling. Aerobic organisms can usually tolerate room air with prompt delivery, but anaerobes are sensitive to oxygen exposure during transit. Use an anaerobic transport system for the portion destined for anaerobic culture, otherwise you risk false negatives.
Do oxygen-requiring organisms always cause infection at oxygen-rich body sites?
They usually prefer those niches, but distribution can also be driven by nutrients, host barriers, and immune defenses. For example, an obligate aerobe may survive transiently deeper in tissue if oxygen and nutrients are locally available, even if it does not establish long-term growth there.
What’s a practical way to avoid common mistakes when culturing microaerophiles like Campylobacter?
Don’t use standard “aerobic” incubation as a substitute. Microaerophiles often need a specific low-oxygen environment (commonly near 5% O2 with elevated CO2). Also verify that plates and media are compatible with that gas mixture, since some preparation details can affect performance.
Why is oxygen sometimes toxic to obligate anaerobes even if they can’t use it for respiration?
They often lack the enzyme defenses that detoxify reactive oxygen species. Short oxygen exposure can generate damaging radicals, even without oxygen being used as a terminal electron acceptor, so their vulnerability is about chemical stress rather than “starvation” for O2.
If an organism produces catalase, does that guarantee it is an obligate aerobe?
No. Catalase is a supporting clue, not a standalone identity test. Some organisms with aerobic metabolism show strong bubbling, but catalase activity can vary by species, growth conditions, and test method. Confirm with oxygen-gradient growth behavior and, when needed, controlled oxygen incubation.