Microorganisms need six things to grow: nutrients (especially carbon and nitrogen), water, the right temperature, a workable pH, the correct oxygen environment, and enough physical space. Remove any one of these and growth slows or stops entirely. That's the whole framework, and everything else in microbiology is just filling in the details of what 'right' means for a particular organism.
What Do Microorganisms Need to Grow: Conditions and Nutrients
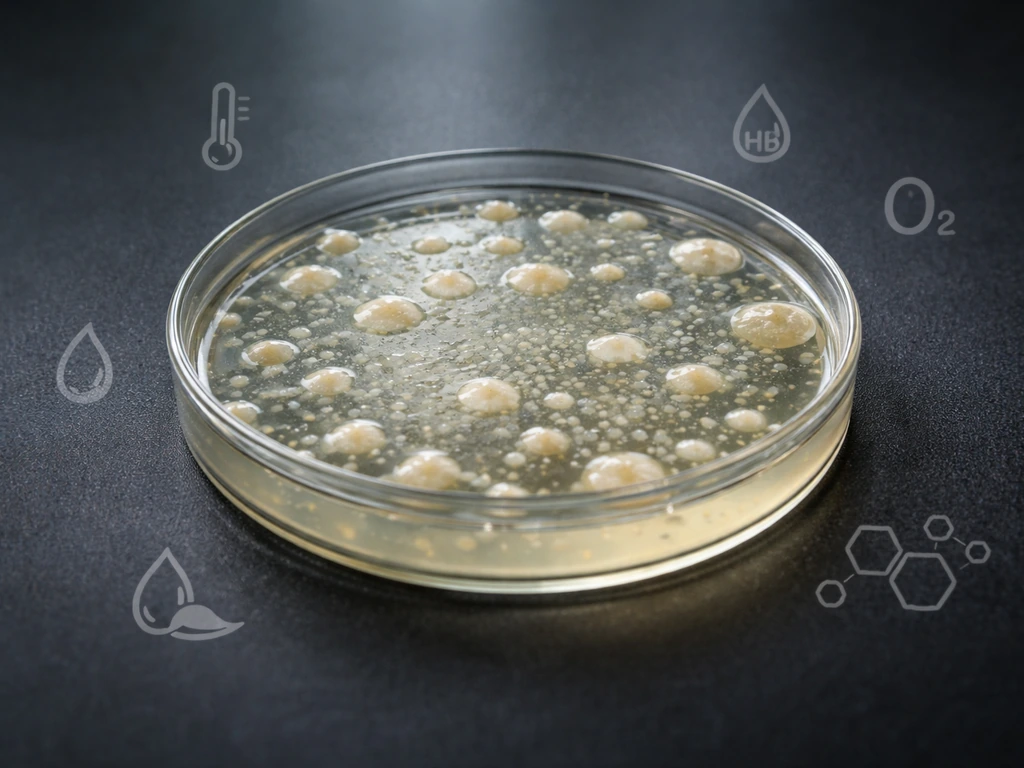
Growing vs. Reproducing: Why the Distinction Matters
When microbiologists talk about microbial 'growth,' they usually mean an increase in the total population, not just a single cell getting bigger. A bacterium grows by taking in nutrients, building new cellular components, and then dividing. So when you ask what microorganisms need to grow, you're really asking: what do they need to keep dividing? If you want the exact breakdown, the key growth requirements like nutrients, water, temperature, pH, oxygen level, and space explain what bacteria need to grow what microorganisms need to keep dividing. This matters because a microbe can survive for a long time without actively reproducing. Bacterial endospores, for example, are the ultimate survival strategy: they drop metabolism to nearly zero, shut down enzyme production, and can resist extreme pH, desiccation, and heat that would kill an active cell. The spore isn't growing, but it isn't dead either. The moment conditions improve, it germinates and starts dividing again. Understanding this dormancy-versus-active-growth distinction is key to making sense of why food safety and hygiene strategies focus on controlling conditions rather than just killing everything.
What Microorganisms Actually Eat: Nutrients Explained

Every living microorganism needs a source of carbon, nitrogen, phosphorus, sulfur, and a handful of minerals. Carbon is the backbone of every biological molecule, from cell membranes to DNA. Nitrogen is essential for building proteins and nucleic acids. Phosphorus shows up in ATP (the cell's energy currency) and in the phosphate backbone of DNA and RNA. Sulfur is needed for certain amino acids, and minerals like potassium, magnesium, calcium, and iron play roles as cofactors for enzymes and structural components.
Where a microbe gets its carbon divides them into two broad camps. Autotrophs, like cyanobacteria, fix carbon from CO2 in the air using light or chemical energy. Most microbes you'll encounter in everyday life, though, are heterotrophs: they need an organic carbon source, meaning they eat sugars, proteins, fats, or other organic compounds from their environment. That's why bacteria thrive on food, skin, and soil, and why a sterile sugar solution with no nitrogen source won't support much growth even though it has plenty of carbon.
Phosphate is worth a special mention because it plays a double role. In lab culture media, phosphate salts are used both as a phosphorus source and as a pH buffer. But add too much and it can actually precipitate out iron and other metals, making them unavailable to the microbe and creating a secondary nutrient limitation. The same principle applies in natural environments: nutrients interact with each other, and a shortage of one can limit growth even when everything else looks fine.
One more thing on nutrients: bacteria can actually acidify their own environment. As they metabolize nutrients, they release organic acids, which lower the surrounding pH. If nothing buffers that change, the pH drop eventually inhibits further growth. This self-limiting feedback loop is one reason fermentation works as a preservation method, and it's a good reminder that nutrient availability and environmental chemistry are never really separate.
The Environmental Conditions Checklist
Temperature

Temperature controls the speed of every enzyme reaction inside a microbial cell. Too cold and enzymes slow to a crawl. Too hot and proteins denature. Every microorganism has a minimum, optimum, and maximum temperature for growth, and these ranges are used to classify them into groups.
| Group | Optimum Temperature | Can Grow At | Example Context |
|---|---|---|---|
| Psychrophiles | ~15 °C or lower | 0 °C or below up to ~20 °C max | Deep ocean, Arctic environments |
| Psychrotrophs | ~20–25 °C | 4–25 °C | Refrigerated food spoilage |
| Mesophiles | ~20–45 °C (often 37 °C) | Moderate room to body temps | Most human pathogens, kitchen bacteria |
| Thermophiles | 50–60 °C and above | High-temperature environments | Hot springs, compost piles |
The practical takeaway: your refrigerator (around 4 °C) does not stop all microbial growth. Psychrotrophs are perfectly happy at fridge temperatures and are the main culprits behind food spoiling in the cold. Freezing (below 0 °C) stops active growth but does not kill most microorganisms, which is why thawed food can go bad quickly once it warms up.
pH
pH affects how proteins fold and function, which means it affects virtually every biochemical process in a cell. Most bacteria prefer a near-neutral pH of around 6.5 to 7.5. Fungi, including yeasts and molds, tend to prefer slightly acidic conditions, roughly pH 5.0 to 6.0, which is one reason you often see mold on fruit before bacteria take over.
Here's something most people get wrong: external pH and internal pH are not the same thing. Bacteria actively pump protons to maintain cytoplasmic pH within a narrow, workable range, even when the environment outside is more acidic or alkaline. Extreme acidophiles (acid lovers) and alkaliphiles (base lovers) have evolved specialized pumps to keep their insides stable under conditions that would destroy a normal cell. This means that adding acid to food, like in pickling, works not by instantly killing bacteria but by making it energetically impossible for them to maintain internal pH homeostasis long enough to grow.
The FDA draws a practical line at pH 4.6 for food safety. Foods with a finished equilibrium pH at or below 4.6 and water activity above 0.85 are classified as acidified foods, while those with pH above 4.6 and water activity above 0.85 are considered low-acid foods and carry a higher risk of supporting dangerous pathogens. That 4.6 threshold is not arbitrary: it's the point below which Clostridium botulinum cannot grow and produce toxin.
Moisture and Water Activity

Water is arguably the most underrated growth requirement. Microorganisms don't just need water to be present: they need free, available water, which is measured as water activity (aw). Water activity is a scale from 0 (bone dry) to 1.0 (pure water), and it measures how much water is actually available to a microbe, rather than the total water content of a food or surface. A bag of sugar can contain water molecules, but those molecules are tightly bound to sugar, leaving an aw too low for most microbes to use.
Most fresh foods have water activity above 0.95, which is plenty to support bacteria, yeasts, and molds. The FDA notes that achieving an aw below 0.85 inhibits growth of microorganisms of public-health significance. Listeria monocytogenes, for example, is considered not able to grow when water activity drops to 0.92 or below in relevant food contexts. Most normal bacteria hit their lower limit around aw 0.91, normal yeasts around 0.88, and normal molds around 0.80. Dried and cured foods exploit this: drop aw below 0.6 and even osmophilic yeasts (which are among the most drought-tolerant microbes) cannot grow. Drying with air and sun is one of humanity's oldest preservation methods, and the microbiology behind it comes down entirely to water activity.
Oxygen: It's Not One-Size-Fits-All
One of the biggest misconceptions I see is the assumption that all microorganisms need oxygen to grow. Some do. Some are actively killed by it. And many don't care either way. The oxygen requirement of a microorganism determines where it can live, what metabolic pathways it uses, and how much energy it can extract from nutrients.
In aerobic respiration, oxygen serves as the final electron acceptor in the electron transport chain, enabling the cell to generate ATP very efficiently. When oxygen isn't available, some microbes switch to anaerobic respiration (using a different final electron acceptor, like nitrate or sulfate) or fermentation (where organic molecules serve as both electron donors and acceptors). Both alternatives generate less ATP than aerobic respiration, which is why anaerobic environments tend to slow growth rates compared to oxygen-rich ones, at least for organisms capable of both.
| Oxygen Category | Relationship with O2 | Where They're Found | Example |
|---|---|---|---|
| Obligate aerobes | Must have oxygen to grow | Air-exposed surfaces, well-oxygenated water | Mycobacterium tuberculosis |
| Obligate anaerobes | Cannot use oxygen; it's toxic to them | Soil, gut, sealed wounds | Clostridium botulinum |
| Facultative anaerobes | Grow with or without oxygen; prefer it with | Almost everywhere | E. coli, Staphylococcus |
| Microaerophiles | Need low O2 (~2–10%) and often elevated CO2 | Mucosal surfaces, microenvironments | Campylobacter jejuni |
| Aerotolerant anaerobes | Tolerate oxygen but don't use it | Gut, fermented foods | Lactobacillus species |
Microaerophiles deserve special attention because they're often misunderstood. They're not just 'anaerobes that can handle a little oxygen.' They genuinely require some oxygen for growth but are damaged by normal atmospheric concentrations (about 21%). Campylobacter, a common food-borne pathogen, is a microaerophile, which is one reason it's tricky to culture in a standard lab setting without specialized equipment.
Bacteria vs. Fungi: Different Organisms, Different Rules

It's tempting to treat all microorganisms as basically the same, but bacteria and fungi are fundamentally different in ways that change their growth requirements and strategies. Bacteria are prokaryotes: single-celled, no nucleus, and they absorb nutrients directly across their cell membrane. Fungi are eukaryotes with nuclei, and they grow primarily as networks of filaments called hyphae (collectively a mycelium) that secrete digestive enzymes into their surroundings and then absorb the breakdown products. This is why fungi are so effective at colonizing solid substrates like bread, wood, and decaying plant matter: they essentially digest the surface from the outside before absorbing the nutrients.
Temperature preferences also differ. Bacteria tend to dominate at neutral to slightly alkaline pH and across a wide temperature range depending on species. Fungi generally prefer slightly acidic pH (around 5.0 to 6.0) and are often more tolerant of dry conditions than bacteria: normal molds can still grow down to aw 0.80, whereas most normal bacteria need at least 0.91. This is why you're more likely to find mold on bread or dried fruit than bacterial colonies.
Some fungi also display a fascinating morphological switch depending on environmental conditions. Certain pathogenic fungi can shift between a mold form and a yeast form based on temperature, pH, and the presence of specific chemicals. This flexibility makes them adaptable to very different environments, including inside the human body. Bacterial growth requirements are covered in more detail in the related topic on what bacteria need to grow, and if you're wondering specifically about fungi versus bacteria when it comes to light, the topic on whether bacteria need light to grow addresses that directly.
How to Figure Out What's Missing (or What's Enabling Growth)
Whether you're trying to prevent mold in your kitchen or understand why a bacterial culture isn't growing in a lab, the troubleshooting process is the same: work through the checklist systematically.
- Check water activity first. Is the surface or food moist? Most spoilage and contamination problems start here. Fresh meat, produce, and cooked foods all have aw above 0.95, which is firmly in the growth-supporting range. If the environment is dry (think crackers, dried beans, or a clean dry countertop), growth is already highly restricted.
- Check temperature. Is the environment in the growth range for the organisms you're worried about? Room temperature (around 20–25 °C) is comfortable for mesophiles and psychrotrophs alike. Refrigeration slows mesophiles but won't stop psychrotrophs. If you're seeing spoilage in the fridge, that's your answer.
- Check pH. Is the environment acidic, neutral, or alkaline? Most bacteria thrive between pH 6.5 and 7.5. Foods or surfaces with pH below 4.6 are hostile to most pathogens. If you're dealing with mold and not bacteria, remember fungi are more acid-tolerant and may keep growing where bacteria can't.
- Check oxygen availability. Is this a sealed container, a deep wound, the center of a thick food product, or an open surface? Obligate anaerobes need sealed, oxygen-free environments. If you're worried about botulism in home canning, it's because sealed low-acid canned foods create exactly the anaerobic, low-acid conditions C. botulinum loves.
- Check nutrient availability. Is there a carbon and nitrogen source available? Sugars, proteins, and fats all serve as carbon sources. A sterile surface with no organic material will not support heterotrophic growth even if temperature, pH, and moisture are perfect.
- Consider dormancy. If conditions are currently poor but were recently favorable, spores or dormant cells may be present and waiting. Improving conditions (warming up, adding moisture, raising pH) can trigger germination and a sudden growth surge.
The same logic applies in reverse if you're trying to understand where and why growth is happening unexpectedly. Trace the conditions back. A bathroom with persistent mold has moisture (aw well above 0.80), an organic substrate (soap residue, dead skin cells), a temperature in the fungal growth range, and probably slightly acidic or neutral pH. Address the moisture and you address the root cause. Scrubbing the visible mold without fixing the humidity just gives it time to regrow.
The broader point is that microbial growth is not random or mysterious. It's predictable once you understand the requirements. Every environment either meets those requirements or it doesn't, and if growth is happening somewhere you don't want it, one or more conditions are in the microorganism's favor. Adjust even one critical factor outside the viable range and growth stops. That's the practical power of understanding what microorganisms actually need.
FAQ
If I change only one condition, will microorganisms always stop growing immediately?
Not necessarily immediately. Some microbes, especially those capable of stress responses, can continue limited growth for a short time after one factor shifts, while they adjust metabolism. Also, changing temperature or pH can trigger slower growth rather than a full stop if you move the environment from optimum toward suboptimal ranges.
How can there be growth when nutrients seem scarce, like on dry surfaces?
Dryness is measured by water activity, not total dryness. A surface can feel dry but still have local micro-wet spots, sweat residues, or trapped humidity (raising aw locally), which provides enough available water for growth. That is why addressing moisture and residues together often works better than just wiping.
What does it mean for oxygen to be “required” if some microbes grow without it?
“Oxygen requirement” refers to the organism’s ability to use oxygen as the final electron acceptor or whether oxygen is tolerated or harmful. Aerobes will fail in strictly oxygen-free environments, anaerobes may grow only without oxygen, and microaerophiles need oxygen below atmospheric levels, so the same “air exposure” can help some microbes and inhibit others.
Why do microbes sometimes grow in the presence of preservatives, even when they should be inhibited?
Preservatives work only when they reach effective conditions at the microbial cell. If the product has high buffering capacity (for example, high phosphate or other buffers), the preservative may not achieve the right pH or chemical form. Also, if water activity is high or nutrients are abundant, microbes can sometimes outlast sublethal inhibition until conditions stabilize.
Does “sterile” guarantee no microbial growth later?
Sterile means no living microbes at the moment of application, but it does not prevent later contamination. Microbes can reintroduce nutrients and water and grow once they enter the environment that meets their other requirements, so storage hygiene and preventing moisture and nutrient sources matter as much as initial sterilization.
Why might a bacterial sample show no colony growth on a culture plate, even if bacteria are present?
Some bacteria can be dormant (for example, endospores) or stressed by harsh conditions, so they may not form colonies immediately. Switching to appropriate recovery conditions, using the correct medium, and controlling temperature, oxygen, and pH for that organism can be necessary to resuscitate growth rather than concluding the sample is clean.
How do autotrophs fit the “nutrients needed to grow” question?
They still require nutrients, but their carbon source is different. Autotrophs use CO2 as the carbon feedstock, then use an energy source like light or chemical reactions, so the “carbon source” requirement depends on whether the organism is autotrophic or heterotrophic.
Is freezing a reliable way to stop all microbial activity?
Freezing typically stops active growth, but it does not reliably kill all microorganisms, particularly those that tolerate freezing and drying. Once the temperature rises during thawing, surviving cells can resume metabolism and division quickly, so safe handling is still needed after thaw.
Why does internal pH matter so much in pickling or acidified foods?
Many microbes can survive outside acidic conditions by actively regulating internal pH using proton pumps. Acidified approaches work when the outside conditions make it too energetically costly for the organism to maintain cytoplasmic pH long enough to divide, not when acid simply acts like an instantaneous poison.
What’s the practical difference between water content and water activity when troubleshooting mold or spoilage?
Water activity (aw) reflects how much water is available for microbial metabolism. A product can have significant total moisture but still have low aw due to dissolved solutes or binding, which can prevent growth. In contrast, small amounts of freely available water can support growth even if the bulk seems dry.




