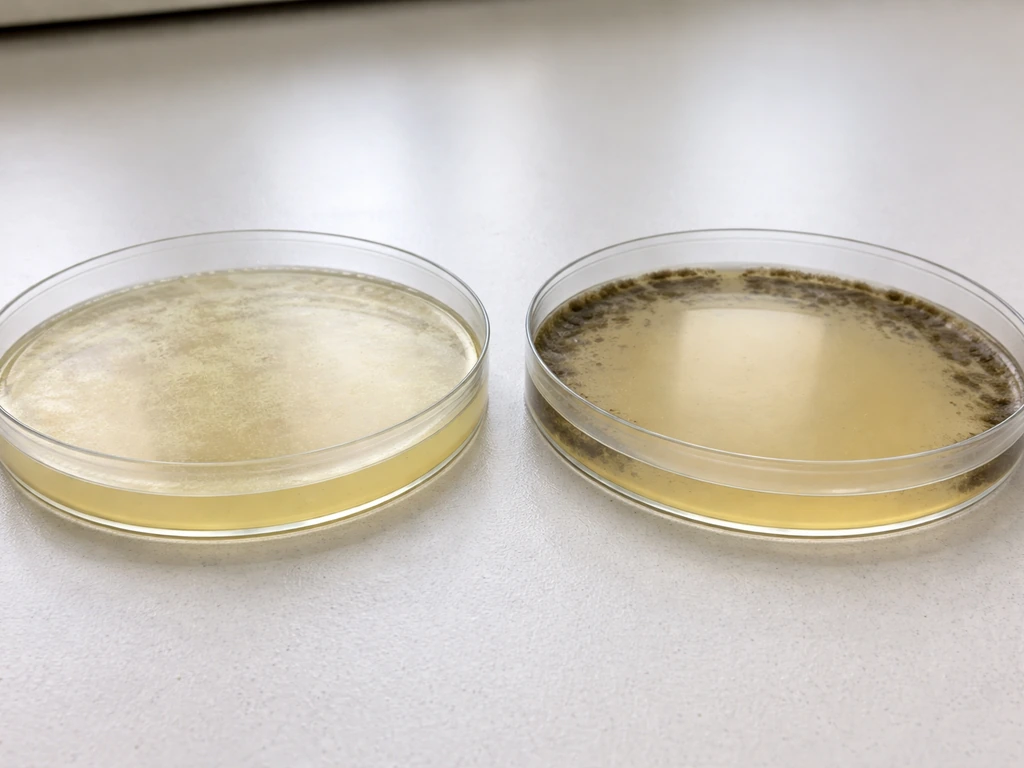
Only transformed bacterial cells grow on the selective agar plate that contains the antibiotic matching the resistance gene on their plasmid. If your plasmid carries an ampicillin-resistance gene (bla), you plate on LB agar with ampicillin (typically 100 µg/mL) and only the cells that successfully took up and expressed that plasmid will survive. Untransformed cells hit that antibiotic and die. That one plate, prepared correctly with the right antibiotic at the right concentration, is your answer.
On Which Petri Dish Do Only Transformed Cells Grow?
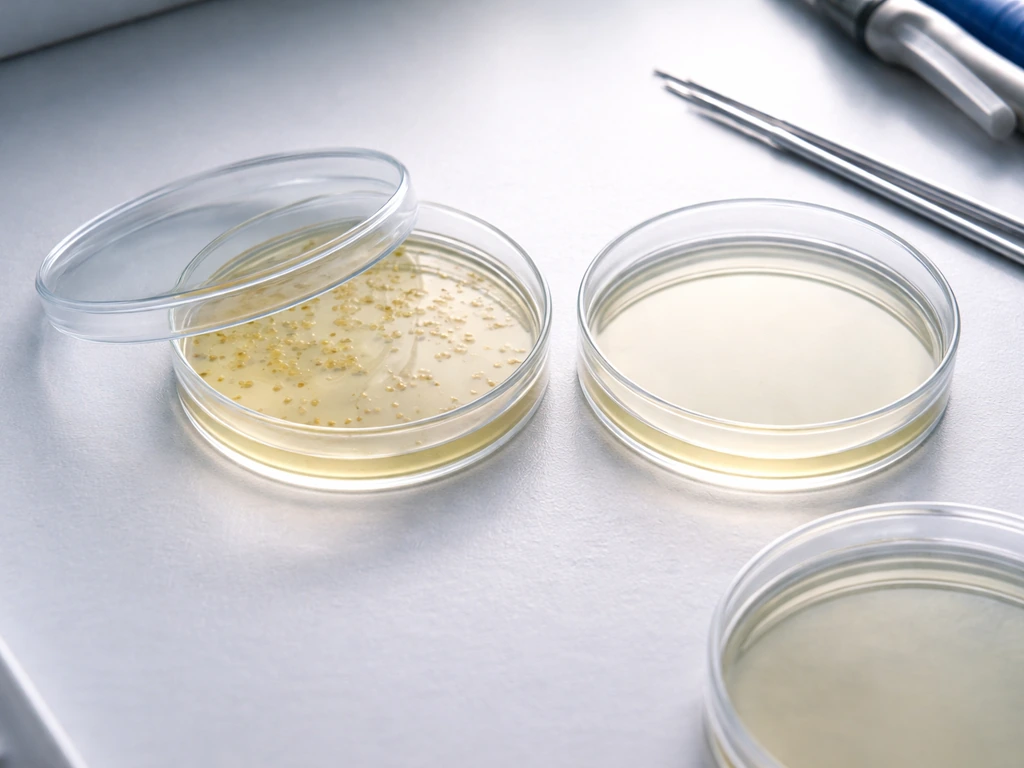
What 'transformed cells' usually means in a petri dish selection experiment
In virtually every undergraduate or high school molecular biology lab, 'transformed cells' means bacteria (almost always E. coli) that have been induced to take up a small circular piece of DNA called a plasmid. The plasmid is engineered to carry a selectable marker, which is almost always an antibiotic-resistance gene. The idea is elegant: if a cell successfully incorporates and expresses the plasmid, it gains the ability to survive an antibiotic that would normally kill it. This is the core principle behind selective plating.
It is worth noting that 'transformation' has other meanings in biology. In oncology it refers to normal cells becoming cancerous, and in some fungal or broader microbiology contexts it can describe other genetic changes. But in the standard lab or classroom context involving petri dishes and selection media, you are almost certainly dealing with bacterial plasmid transformation. That is the interpretation this guide focuses on, and it is the scenario where the selective agar principle applies directly.
Why transformed cells grow: matching the plasmid's resistance marker to the right medium
Here is the underlying logic, because understanding the 'why' saves you a lot of troubleshooting headaches later. A plasmid carrying the bla gene produces beta-lactamase, an enzyme that physically breaks down ampicillin in the surrounding medium. A cell that has that plasmid can degrade the antibiotic around it and grow freely. A cell without the plasmid cannot neutralize the antibiotic, so it cannot replicate. The agar plate with ampicillin is not just a filter; it is an active selection pressure that kills cells lacking the resistance gene while rewarding cells that have it.
The same logic applies to every other common resistance marker. Kanamycin resistance (the nptII or KanR gene) produces an aminoglycoside phosphotransferase that inactivates kanamycin. Chloramphenicol resistance (CamR/cat) produces chloramphenicol acetyltransferase. Tetracycline resistance (TetR/tet) works through efflux pumps. Each resistance mechanism is specific to its antibiotic, which is why you cannot substitute one antibiotic for another and expect the same selection to work.
Which petri dish to use: the selective agar matched to your plasmid
The single most important step before you ever touch a plate is to look at your plasmid map or protocol sheet and identify the resistance gene. This sounds obvious, but using the wrong antibiotic plate is one of the most common practical mistakes in transformation labs. Once you know the marker, you pick the agar containing that antibiotic at the appropriate working concentration.
| Resistance Marker | Antibiotic to Use | Typical Working Concentration (µg/mL) | Notes |
|---|---|---|---|
| bla (AmpR) | Ampicillin (or carbenicillin) | 25–100 (ampicillin); 50–100 (carbenicillin) | Carbenicillin is more stable; reduces satellite colonies |
| nptII / KanR | Kanamycin | 10–50 | 50 µg/mL is a common teaching-lab standard |
| cat (CamR) | Chloramphenicol | 25–170 | Wide usable range; check your specific protocol |
| tet (TetR) | Tetracycline | 10–50 | Light-sensitive; prepare plates in subdued light |
A common teaching-lab standard that works well for most AmpR plasmids is 100 µg/mL ampicillin in LB agar. For KanR plasmids, 50 µg/mL kanamycin is a reliable starting point. These concentrations are not arbitrary: too low and you get background growth from untransformed cells; too high and even transformed cells can be stressed. The ranges above reflect guidance from multiple protocols used in molecular biology teaching labs.
Beyond the antibiotic concentration, the physical condition of the plate matters more than most students realize. Antibiotic-containing plates should be stored at 4°C and used within about 4 weeks of preparation. Plates that have been sitting at room temperature for weeks can have degraded antibiotic, which will ruin your selection. Before you plate, inspect the agar surface: it should be smooth, moist but not wet, and free of condensation on the surface. Bring plates to room temperature or briefly warm them (around 37°C for 15–30 minutes) before plating to help even spreading and reduce condensation.
Controls you need and how to read your results
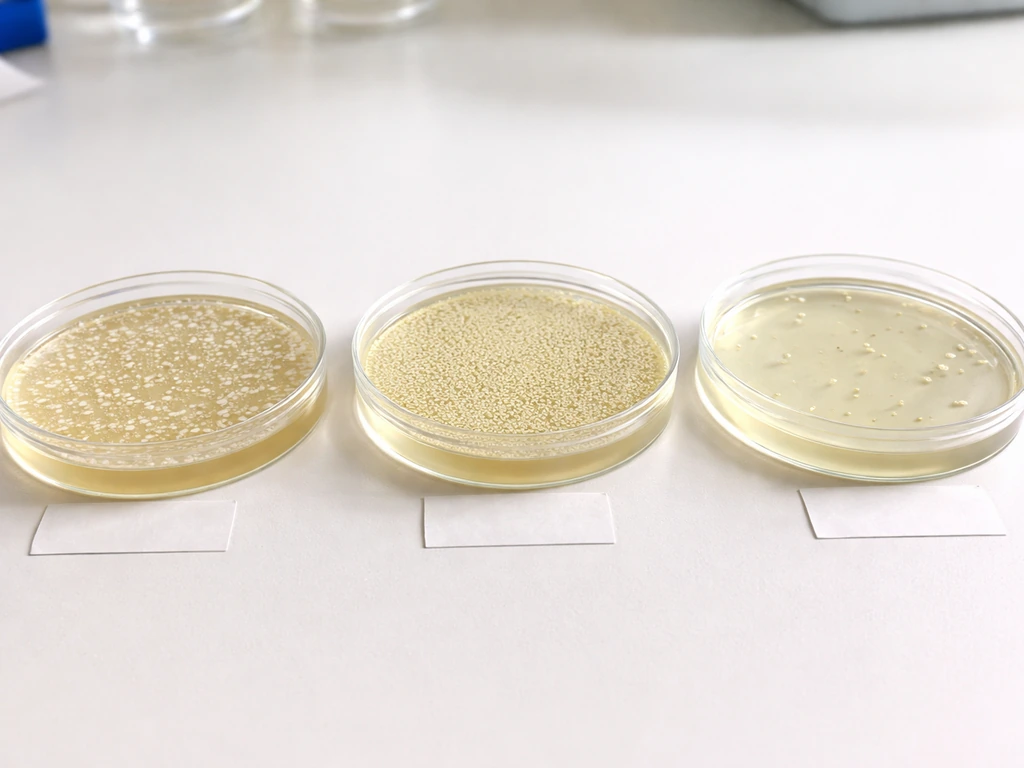
Running controls is not optional extra work; it is how you know whether your results are real. Without controls, a plate full of colonies could mean successful transformation or it could mean your antibiotic degraded and every cell survived regardless. Here are the three controls that matter:
- Positive control: Take cells you know are already resistant (or use your transformation mix with a known plasmid) and plate on the selective agar. You should see robust growth. If you do not, something is wrong with your plate or your cells.
- Negative control (no-DNA control): Put your competent cells through the entire transformation procedure but add water instead of plasmid DNA. Plate on the same selective agar. You should see no growth, or at most a very tiny number of background colonies. Growth here tells you the antibiotic is not working.
- Untransformed cell control on non-selective agar: Plate untransformed cells on plain LB (no antibiotic). You should see a lawn of growth. This confirms your cells were viable going into the experiment.
Reading your results comes down to this pattern: colonies on the antibiotic plate from your transformation reaction indicate successful selection of transformed cells. No colonies on that same plate from your no-DNA control confirms the antibiotic is active and the selection is real. If both the transformation reaction and the no-DNA control show colonies, your antibiotic has failed and your results are meaningless until you troubleshoot the plate conditions.
Common failure modes: satellite colonies, background growth, and what to do
Satellite colonies are the most frustrating result in ampicillin selection, and they trip up students and experienced researchers alike. They appear as tiny, hazy rings of smaller colonies surrounding a larger, true transformant colony. Here is what is happening: the true transformant expresses beta-lactamase, which leaks into the surrounding agar and locally degrades the ampicillin. Once that local antibiotic concentration drops low enough, nearby untransformed cells can survive and form those small satellites. The satellite colonies do NOT contain your plasmid, even though they are on your selective plate.
Several factors make satellite colonies worse: plates that are old (antibiotic has partially degraded before you even plate), over-plating too many cells at once (more beta-lactamase activity faster), and incubating too long (more time for antibiotic to degrade). The practical fixes are to use carbenicillin instead of ampicillin (it is a more stable beta-lactam antibiotic and dramatically reduces satellite formation), use fresh plates, do not over-plate, and pull plates after 14–18 hours rather than letting them sit longer.
Background colonies on plates that should show no growth (your no-DNA control) point to a different problem: antibiotic failure. The most likely causes are degraded antibiotic in old or improperly stored plates, antibiotic that was added to agar that was too hot (above 55°C) when plates were poured (heat destroys many antibiotics), or the wrong concentration was used. Check your plate preparation records. If in doubt, make fresh plates and rerun your controls before repeating the full experiment.
- Satellite colonies around large colonies: switch to carbenicillin, use fresher plates, reduce cells plated, shorten incubation
- Background growth on no-DNA control plate: antibiotic likely degraded; remake plates with fresh antibiotic stock
- No colonies anywhere, including positive control: competent cells may be dead; check heat shock conditions and cell viability
- Colonies on selective plate but also on no-DNA control: antibiotic concentration too low or plate too old; do not trust these results
- Fewer colonies than expected: competent cell efficiency may be low, or recovery time after heat shock was too short
Incubation conditions that make or break your selection

After plating, invert your plates (agar side up) and incubate at 37°C, the standard temperature for E. coli growth. Most teaching protocols call for 14–18 hours of incubation, and a total of 16–20 hours is a reasonable window if colonies are slow to appear. Pulling plates at the shorter end of that range (around 14–16 hours) is actually better for ampicillin selection because it limits the time available for beta-lactamase to degrade the antibiotic and generate satellites.
Temperature also matters for a subtler reason: antibiotic stability. Ampicillin and other beta-lactams are not stable indefinitely even at 37°C. The longer your plate sits warm, the more antibiotic breaks down. This is why leaving plates in the incubator for 36 or 48 hours trying to coax out more colonies is counterproductive with ampicillin selection; by that point the selection may have broken down completely, and any colonies appearing late are not trustworthy. Kanamycin and chloramphenicol are more stable in this regard, making them somewhat more forgiving for selection timing.
One step that is easy to skip but genuinely improves results is the recovery incubation before plating. After heat shock (the step that briefly stresses cells to allow plasmid uptake), cells are usually incubated in plain, antibiotic-free liquid media (SOC or LB) at 37°C for 45–60 minutes with shaking. This recovery period gives the transformed cells time to start expressing their new resistance genes before they hit the antibiotic plate. If you skip or shorten this step, even successfully transformed cells may not yet be expressing enough resistance protein to survive, and you will get fewer or no colonies on your selective plate.
Your practical next steps, right now
If you are solving this today, here is exactly what to do. First, open your plasmid map or protocol and find the resistance gene listed under selectable markers. Common names to look for are bla (ampicillin/carbenicillin), KanR or nptII (kanamycin), CamR or cat (chloramphenicol), and TetR (tetracycline). Second, confirm you have agar plates containing that antibiotic at the correct working concentration, prepared within the last 4 weeks and stored at 4°C. Third, set up your three controls (positive, no-DNA, and non-selective) alongside your experimental plates. Fourth, incubate inverted at 37°C and check at 14–16 hours. Only the plate combining the right antibiotic at the right concentration with your transformed cells should show robust, evenly distributed colonies.
Understanding the conditions that allow only transformed cells to grow is really about understanding the biology: the resistance gene on the plasmid produces a specific enzyme or protein, and only the agar plate that challenges cells with the matching antibiotic will reveal which cells have that protection. Everything else, controls, concentrations, incubation time, is about making sure that biological signal comes through cleanly. This same principle of selective media for specific microbial growth shows up across many areas of microbiology, from enrichment cultures designed to grow specific microbial populations to the broader question of what environmental conditions allow any microorganism to thrive or stay dormant. In enrichment cultures, you choose the media type that supports the specific microbes you want to grow. Single-celled microorganisms can form irregular colonies or masses when they are able to grow under the right environmental conditions. Once you understand which selective condition matches your cells, you can decide what you can grow in a petri dish what can you grow in a petri dish. Growing cells in a petri dish depends on choosing the right selective medium and incubation conditions how to grow cells in a petri dish.
FAQ
Is it always only one specific petri dish where only transformed cells grow?
Usually, no. The answer depends on which resistance marker is on the plasmid. A single selective plate will allow only cells that carry and express that specific marker to grow, while cells without the plasmid are inhibited. If you plate on the “wrong” antibiotic (or wrong concentration), non-transformed cells may survive, or transformed cells may be killed.
What could cause untransformed cells to grow even when I used the correct antibiotic?
A plate with the correct antibiotic can still produce colonies from non-transformed cells if the antibiotic is degraded, too low, or was added after the agar cooled below the intended range. This is why the no-DNA control must be clean, and why stale plates, improper storage, or incorrect pouring conditions are common causes of misleading growth.
If I see colonies on the ampicillin plate, does that mean every colony is a transformed cell?
The “only transformed cells” claim applies to the selection strength of the antibiotic, not to the plasmid status of every colony. On ampicillin plates, satellite colonies can form around true transformants after local breakdown of ampicillin. Those satellites often do not carry the plasmid, so you should pick well-isolated primary colonies for downstream work.
Why can I get zero colonies on the selective plate even if transformation worked?
Yes, plasmid expression matters. If you plate immediately after heat shock without sufficient recovery in antibiotic-free media, transformed cells may not yet produce enough resistance protein to survive the antibiotic challenge, leading to false “no colonies” or very low numbers.
When should I add the selective antibiotic relative to heat shock and recovery?
You typically use the selective agar only after the recovery period. After transformation, cells are usually given 45–60 minutes in SOC or LB (no antibiotic) to initiate resistance gene expression, then spread onto the selective plate. Skipping this step is a common reason selection appears to fail.
What should I do if my no-DNA control plate also shows colonies?
If both the experimental transformation plate and the no-DNA control show growth, treat the result as an antibiotic failure. Do not trust colony counts, and instead remake plates with fresh antibiotic, confirm the working concentration, and verify plate storage and pouring conditions (especially that antibiotics are not overheated during gel or agar preparation).
Why is reading colonies at the correct time important for distinguishing true transformants from artifacts?
Yes, colony timing affects reliability, especially with ampicillin. Checking too late can increase satellite and background growth because beta-lactamase accumulates and the antibiotic breaks down over time. A practical approach is to score colonies around the expected 14–18 hour window rather than waiting for “more colonies.”
How does the amount of plated cells affect colony quality and satellite colonies?
Over-plating can worsen satellite formation because many transformants produce more enzyme quickly, accelerating local antibiotic degradation. If you see lots of satellites or a lawn-like appearance, repeat with a lower cell volume or dilution that yields well-spaced colonies for picking.
Are incubation time limits different for ampicillin versus kanamycin or chloramphenicol?
Some antibiotic systems are more stable than others, so the optimal incubation window can differ. For example, beta-lactams like ampicillin are less forgiving at warm temperatures, while aminoglycoside and chloramphenicol selections are often more stable, making them less prone to selection breakdown late in incubation.
What’s the safest way to choose which selective agar to use if the plasmid map is unclear?
If you are unsure what marker your plasmid has, you cannot reliably choose the correct selective plate. The selection marker is defined by the plasmid map or protocol, names like bla, nptII, cat, or tet, and sometimes specific brand naming. Verify the resistance gene before selecting the agar, otherwise you risk false negatives or false positives.